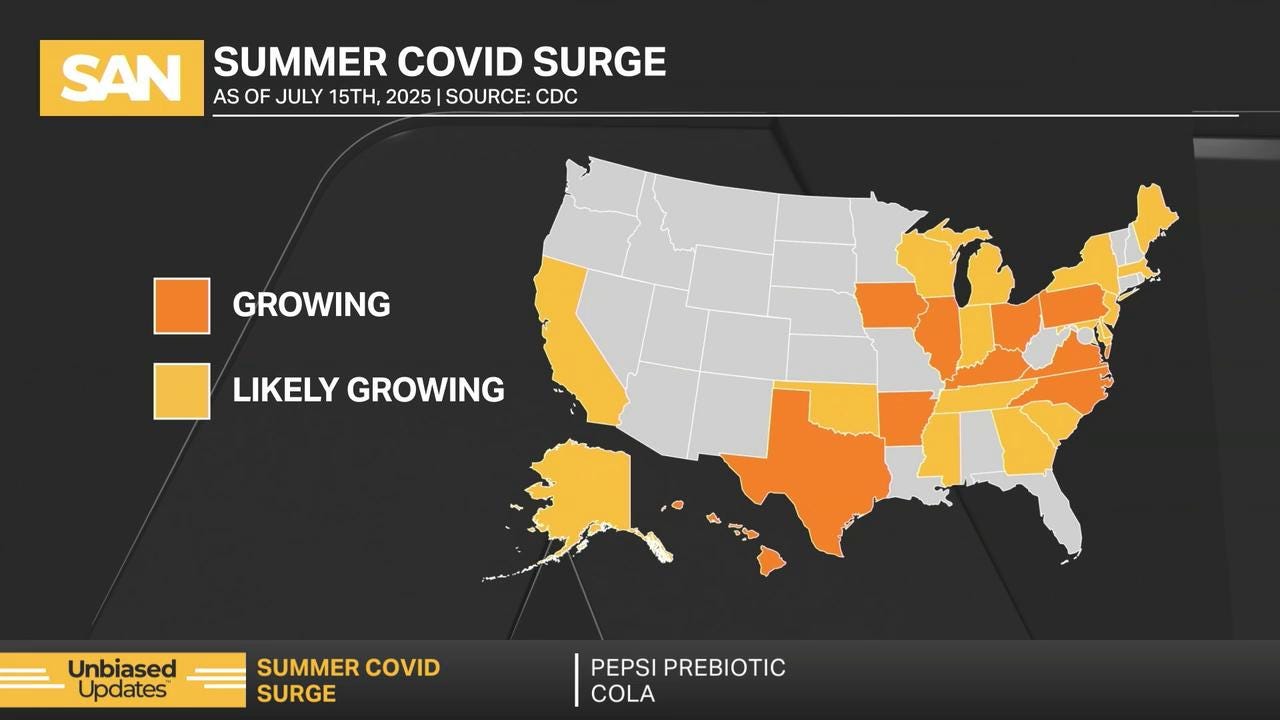
CDC: 26 states have normal summer waves spreading
CDC data shows that there are rising Covid-19 cases in more than 26 states, with mild new variants and latest vaccine guidance for adults and children.
Straight Arrow News
Health and Human Services Director Robert F. Kennedy Jr. has revealed that his agency cut funds for mRNA development and calls vaccine technology “ineffective” and claims it poses more risk than profits.
X In a video posted on August 5th, Kennedy said the decision would affect 22 projects worth nearly $500 million in the Bureau of Advanced Biomedical Research and Development, which will help businesses develop medical supplies to address public health threats.
Messenger RNA, or mRNA, is used in two of the most common COVID-19 vaccines approved in the announcement of the US Kennedy.
“We’ve seen a lot of different things,” said Jeff Coller, professor of RNA biology and therapy at Johns Hopkins University. “As other countries advance these proven safe and effective treatments, American patients may increasingly rely on foreign innovations for groundbreaking treatments.”
How do mRNA vaccines work?
The Covid-19 vaccine works by instructing the body’s immune system to recognize the virus and creating antibodies that attack it.
Messenger RNA, or mRNA, is a code that tells the body’s cells to produce only parts of the virus, proteins on the surface. The cord is protected by a fat foam-like lipid coating.
When injected into the body, the vaccine releases mRNA and programs the cells to produce spike proteins like those found on the surface of the Covid-19 virus SARS-Cov-2. Our immune system recognizes these vaccine-created spike proteins as invaders and creates antibodies that block future attacks from the virus.
Messenger RNA vaccines contain only a small portion of the virus, and unlike some vaccines, people cannot be given diseases that prevent or cause allergies to eggs and other traditional vaccine ingredients.
What did RFK say about mRNA?
In a video posted to X, Kennedy claimed that the mRNA vaccine is ineffective against mutated versions of the virus, revealing that HHS plans to redistribut the funds to study a whole virus vaccine that uses weakened versions of the virus.
Kennedy’s video “represents a fundamental misconception of science,” Koller said. Vaccines using mRNA technology are still effective against slightly mutated versions of the virus. Even if mutant viruses can avoid immunity, scientists can still modify the mRNA code to create new vaccines that target mutant proteins more rapidly than other vaccine technologies.
Additionally, Koller said that because vaccines introduce more viral particles into the human body, they tend to produce more side effects than other types of shots, which are protein-based or mRNA. There is also a slight risk that a person can get sick due to a disease that the vaccine is intended to prevent.
What does mRNA technology refund mean?
The most obvious result of reimbursing mRNA vaccine development is the loss of resources to improve vaccine technology targeting infectious diseases such as Covid-19, avian influenza and cancer, Coller said.
But he also said that while research could be paid without federal support, he would send a “terrifying” message to vaccine companies about continuing this type of research in the United States.
This could allow businesses to move their operations and marketing to other countries. This not only makes these treatments more expensive in the US, but also replaces them as “the world leader in biotechnology,” Koller said.
“The rest of the world has doubled mRNA drugs. That’s true and the US will lose race,” Koller said. “The decision from Kennedy is essentially destroying our control in the field of biotechnology.”
Should we expect a Covid vaccine this fall?
Dr. Beth Oller, a family doctor in Stockton, Kansas, and Dr. Beth Oller, a clinical instructor at the University of Kansas School of Medicine, said it is unclear whether patients should expect a Covid-19 vaccine this fall.
In June, Kennedy fired all members of the Advisory Committee on Vaccination Practices. This is an important vaccine panel that makes recommendations for Covid-19 and other vaccines, and has appointed eight new members. The committee did not make any autumn recommendations during its first meeting, and is not expected to resume until “September/October,” according to the Centers for Disease Control and Prevention’s website.
Kennedy said on May 27 that the Covid-19 vaccine will no longer be included in the CDC’s recommended vaccination schedule for healthy children and pregnant women.
Under the change, the only people recommended for the Covid-19 vaccine are those over the age of 65 and those with existing health problems. This could make it difficult for others who want the Covid-19 vaccine to get it, such as healthy people under the age of 65 with vulnerable families or those who want to reduce the short-term risk of infection.
The American Obstetricians and Gynecologists (ACOG) and the American Academy of Pediatrics (AAP) have issued statements, among other organizations, condemned the change.
Does insurance cover my covid shot?
A recent poll shows that many consumers are worried about the possibility of reducing health insurance companies’ vaccine coverage.
A KFF poll issued on August 1 found that 40% of adults said they would “undoubtedly” or “probably” get a shot of the Covid-19 vaccine. Of those planning on getting the vaccine, 62% said they were worried that their insurance wouldn’t cover the costs of the vaccine.
The group representing commercial health insurance companies said it plans to cover vaccines recommended by the vaccine advisory panels CDC and ACIP.
“The health plan will continue to follow federal requirements related to coverage of ACIP recommended vaccines and will continue to support broad access to important preventive services, including vaccinations,” said Tinastow, spokesman for AHIP, representing health insurance companies.
The CDC currently recommends shared decisions between parents and doctors to vaccinate children with the Covid-19 vaccine. It also recommends vaccines to people with moderate or severe immunity compromised.
James Gelfund, president and CEO of the ERISA Industrial Committee, said large employers who provide health insurance benefits to workers and their families are likely to continue to cover the Food and Drug Administration-approved vaccines.
In some cases, employer vaccine coverage may follow FDA approval rather than narrowing ACIP recommendations.Contribution: Mary Wallas Holldridge
Adrianna Rodriguez can visit adrodriguez@usatoday.com.

